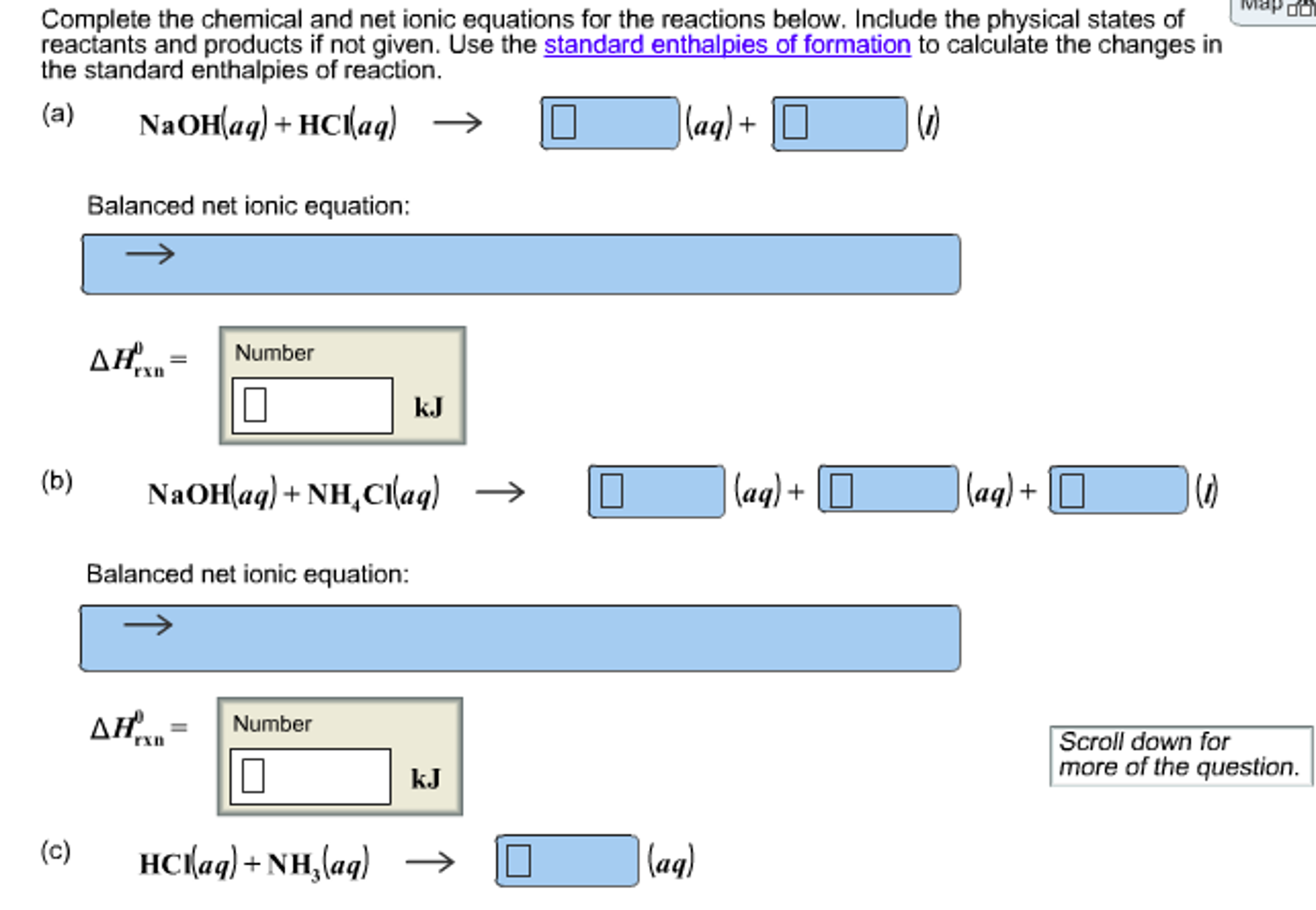
  So our 1st 1 is this complex ion of iron, shes going to act as a weak acid. Educators earn digital badges that certify knowledge, skill, and experience. Add the number of electron lost and gained by each part such as. VIDEO ANSWER: So were going to write some net ionic equations here with the ions that were given are going to react with hydroxide. Save time lesson planning by exploring our library of educator reviews to over 550,000 open educational resources (OER).Īn all-in-one learning object repository and curriculum management platform that combines Lesson Planet’s library of educator-reviews to open educational resources with district materials and district-licensed publisher content.Ī comprehensive online edtech PD solution for schools and districts. Timely and inspiring teaching ideas that you can apply in your classroom Manage saved and uploaded resources and foldersīrowse educational resources by subject and topic Please let us know how we can improve this web app.Search reviewed educational resources by keyword, subject, grade, type, and more To obtain the net ionic equation for this chemical equation, enter the equation in the prompt box of the net ionic equation calculator. calcium hydroxide + carbon dioxide = calcium carbonate + waterĮxamples of the chemical equations reagents (a complete equation will be suggested): Equation balancer is an online tool for balancing chemical equations.To write ionic equations follow the steps below. While ionic equations show all of the substances present in solution, a net ionic equation shows only those that are changed during the course of the reaction. PhCH 3 + KMnO 4 + H 2SO 4 = PhCOOH + K 2SO 4 + MnSO 4 + H 2O We write net ionic equations to understand what is changing in a chemical reaction. Ionic equations and net ionic equations are usually written only for reactions that occur in solution and are an attempt to show how the ions present are reacting.Find more Chemistry widgets in WolframAlpha. Q: Assume you titrated to the endpoint 0.10 moles of HA (weak, monoprotic acid) with a solution of A: Click to see the answer. A: According to the law of conservation of mass, the quantity of each element does not change in a questionanswer. 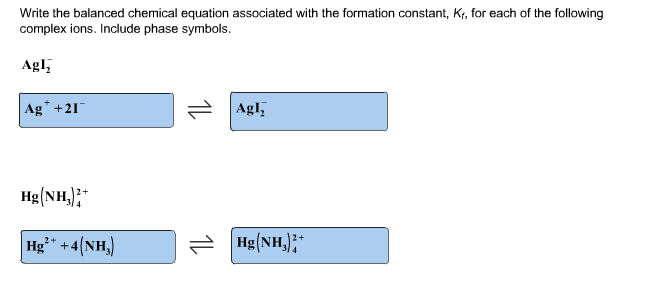
BALANCED NET IONIC EQUATION CALCULATOR FREETo enter an electron into a chemical equation use + H 2O Get the free 'NET IONIC EQUATION CALCULATOR' widget for your website, blog, Wordpress, Blogger, or iGoogle. Complete and balance the following equations then write balanced net ionic equations a.Compare: Co - cobalt and CO - carbon monoxide T/F False All net ionic equations must be balanced by both mass and charge. T/F False Spectator ions undergo changes in a chemical reaction. T/F False Spectator ions are included in net ionic equations. Always use the upper case for the first character in the element name and the lower case for the second character.Ğxamples: Fe, Au, Co, Br, C, O, N, F. The net ionic equation for the reaction between aqueous solutions of HF and KOH is The net ionic equation for the reaction between aqueous solutions of HF and KOH is. ions Spectator ions can be included as part of a net ionic equation.Redox reactions are commonly run in acidic solution, in which case the reaction equations often include H 2 O(l) and H + (aq). Follow the given process to use this tool. 
Write the molecular equation ionic equation and net ionic equation for the reaction of aluminum bromide and silver hydroxide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
